Samuelson gave an Institute Colloquium recently in which he discussed his work on copper complexes, and why they matter

Ashoka G Samuelson is a professor at the Department of Inorganic and Physical Chemistry, IISc. He joined the Institute after completing his PhD from Cornell University, USA, in 1983. His research is centred on understanding how a chemical reaction proceeds before it reaches the product. More recently, his team has been exploring the applications of metallic complexes, particularly copper complexes, with suitable ligands in treating cancer.
On 30 March 2017, Samuelson delivered an Institute Colloquium, after which he sat down with Connect to talk about his journey as a chemist.
Can you tell us about your early life? When did you realise you wanted to become a chemist? What drew you to this field?
After my Pre-University degree, I had to make a choice for my Bachelor’s. I tried my best to move away from chemistry; I did not want to be labelled as someone who became a chemist due to parental influence. But I realised that only chemistry permitted endless creativity. It is still true. Chemistry is a creative science.
Can you tell us about your life as a PhD student at Cornell University? What did you work on?
I worked on a physical organometallic chemistry problem. Physical organic chemistry is still my first love. Much of my work still involves understanding reactions through the use of isotopes. However, that part of my research is more involved and would be difficult for a general audience to follow. The papers read like detective stories where we try to figure out the path taken by the molecules during a reaction.
Can you tell us about how you set up your lab after taking charge?
Slowly, and with great difficulty! It took some time to obtain a research grant. In those days, the funding agencies did not have the same magnanimity they have now. We set up a lab to handle air and moisture sensitive compounds, but could not get sufficient funds to import a dry-box.
You’ve been associated with IISc for the last 34 years. How would you describe this journey?
This has been a wonderful experience in my life. I would not trade my position here with a position in any other institution in India or even abroad. The academic atmosphere in our department is wonderful. It would take several pages to describe this journey.
How different is it teaching undergraduate students as compared to teaching PhD students?
I have been teaching undergraduates [UG] for only the past three years. The experience has been improving. The challenge is to convince them that learning the concepts you are trying to teach would have a lasting value even if they are not going to be working in the area of chemistry associated with the course. So I try to teach the logic behind various studies and try to do case histories as well so that the basic principle of scientific research is understood. UG students are more easily excited compared to PhD students. After going through a Master’s programme, there is very little energy left for studying! But then generalisations are difficult.
So I try to teach the logic behind various studies and try to do case histories as well so that the basic principle of scientific research is understood
You started your research here by studying copper complexes. Can you tell us why you chose copper (l) complexes over other metal complexes and also throw some light on other areas of research?
We studied Copper (l) complexes for two reasons – its role in speeding up reactions and carbon dioxide capture. Palladium (0) is an excellent catalyst and we hoped copper (I) could mimic palladium (0) as they have similar electron counts. And we achieved moderate success in this field. We were also interested in looking at how copper (I) could mimic the role of nickel (0), a carbon dioxide activator, as Nickel (0) also has the same electron count.
Another metal complex we used to capture carbon dioxide was titanium tetraisopropoxide. It doesn’t react spontaneously but in the presence of one molecule of water, carbon dioxide capture does occur. But the efficiency wasn’t that good as two titanium tetraisopropoxide molecules were required to capture one molecule of carbon dioxide.
We also worked on understanding on how two copper atoms come close together [a phenomenon called cuprophilicity] despite repulsion from the filled 3d orbitals of each copper (I) atom. During the course of our research, we realised that copper complexes which are soft cations can form more stable complexes in the presence of both hard and soft ligands [referred to as the optimisation principle], which goes against the widely believed principle – soft cations bound to soft ligands would only like to bind soft ligands rather than hard ligands [maximisation principle]. Working on copper complexes turned out to be like working in a gold mine.
Working on copper complexes turned out to be like working in a gold mine
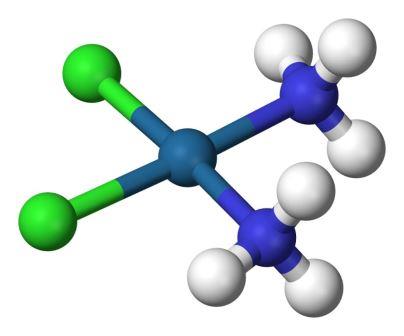
In your talk, you made a mention of the scientific debate on cuprophilicity. Can you explain that?
Gold atoms can come close together, otherwise called aurophilicity, because there is an inherent mixing of 6s and 5d orbitals, bringing the metal atoms together. This is on expected lines. But what came as a surprise was the fact that the copper-copper distance was found to be short in several complexes. There were two conflicting views based on this. One group believed that ligands were responsible for the short distances and another group was of the opinion that metals were making a difference.
So we studied factors that promote short distances. We understood that the ligand was inducing short distances between two copper complexes. Ligands are capable of removing electron density from metals, thereby reducing repulsion and bringing the metal atoms together. It took us about 15 years to arrive at this conclusion systematically and definitively.
Did you experience any eureka moment while working in the lab? Can you tell us about it?
We have had many eureka moments. The identification of orthocarbonates as a product in the carbon disulphide reaction was a startling discovery and our first eureka moment. This was made while we were trying to develop reactions that can capture carbon dioxide. We used carbon disulphide as a model reaction with copper aryloxides. Orthocarbonates were relevant in the 1980s because they were used to make polycarbonates that are widely used in making CDs. Our method of making orthocarbonates was rather convenient as opposed to the earlier more complicated methods. Back then, we didn’t realise the value of orthocarbonates. Later, GE plastics patented a method of conversion of orthocarbonates to polycarbonates and cited our paper as well.
We were the first ones to show the difference in reactivity between two extremes ends of the d block elements. We originally studied copper (d Block element) which is on the right side of the periodic table and also looked at the left side of the d block elements like titanium and zirconium with the help of my students, Rajashekar Ghosh and Akshai Kumar. We were surprised to see the reactivity between the two ends were indeed very different. The difference can be attributed to differences in electron count. The nucleophilicity of the coordinated alkoxide goes up for metals with high electron density while for metals with few electrons; the reactivity is the exact opposite.
The fact that copper (I) prefers hard ligands to soft ligands was another eureka moment too.
Apart from its role in catalysis, do organometallic complexes have any other applications?
These complexes are used increasingly in medicine and in making new materials.
In your talk, you spoke about the utility of another metallic complex, ruthenium half-sandwich complex, in reducing the harmful effects of a drug used to treat blood cancer. How did you go about this?
6 Thioguanine (TG) is used to treat Acute Myeloid Leukemia (AML). Unfortunately, use of 6 TG can lead to skin cancer if the patient is exposed to UVA radiations from the sun. One ends up with skin cancer while treating blood cancer. My student, Raja Mitra, used 6-TG with a ruthenium half-sandwich complex to carry the active molecule to the cancer cell. We used NMR [nuclear magnetic resonance] and mass spectrometry to look for photochemical products that are normally formed when 6 TG undergoes oxidation. Ruthenium (Ru) complexes with 6-TG did not show any such products. This has two advantages. One is that it is transported more efficiently to the cancer cell and another unexpected effect is that it is less reactive to sunlight. We think the use of Ru (6-TG) complex might be useful. But it has to be shown that is more active and less damaging than 6 TG in vivo. A lot more research is required. The Office of Intellectual Property and Technology Licensing (IP TeL) from IISc has contacted several companies. A start-up company will be testing this complex and taking this forward.
The Office of Intellectual Property and Technology Licensing (IP TeL) from IISc has contacted several companies. A start-up company will be testing this complex and taking this forward
You are now focussing on using copper complexes to study anti-cancer activity. Why did you choose Cu (ll) over Cu (l)?
We started our research by testing copper (I) complexes but copper (I) is not suitable for the targeted delivery work. Copper (II) complexes are better suited due to their stability in an aqueous environment; they also have ligands which we can easily attach to macromolecules.
You spoke about triple targeted therapy to reduce tumour burden. Can you explain this?
We explored copper complexes for targeted delivery to reduce tumour size. We used a combined approach of gold nanoparticles carrying metallic complexes and decorated with vitamins. Cancer cells have more receptors for vitamins than normal cells; we used biotin to enable targetted delivery of the drug conjugate. Glutathione (GSH) cleaves disulphide bonds. So we used a disulphide linker with the drug conjugate as the GSH concentration is higher in cancer cells thereby leading to release of the complex from the conjugate. We collaborated with Kumaravel Somasundaram, Professor, Department of Microbiology and Cell Biology, to test this on mice. The results were encouraging as treated mice showed tumour size reduction by 75%.
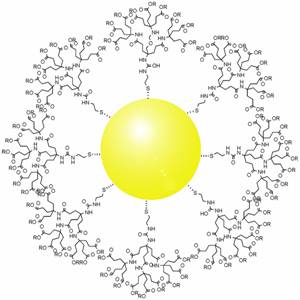
After the success you’ve obtained with copper complexes in mice, what is your next plan of action?
The targeting efficiency has to be improved as we realised that the addition of biotin to the drug complex was only 5-10 % more efficient than the drug without biotin. This is something that we are hoping to explore. One of my students is working on a different method to increase biotin concentration on the drug conjugate to increase the efficiency. I would like to hand over the problem to younger colleagues on this campus or elsewhere because it would take a long time.
What advice would you give to budding chemists?
Ask the right questions! Sounds easy, but framing right questions are tough, especially in today’s context.
I believe one should have two types of research going on. One based on the tough long-term goals and the other based on topical problems which will find acceptance in today’s journal system which relies on catchwords and hype. Unlike most new institutions, IISc should support open-ended problems. Personally, I am in favour of old style freedom to do research without pressure. However, one also has to move with the times.