How researchers at IISc are working on turning research on translation to translational research
When Saumitra Das was a postdoc at the University of California, Los Angeles, he discovered how to inhibit polio virus translation. That is, how to stop the virus from hijacking its host cell’s machinery to manufacture its own proteins. Das had used a small RNA derived from yeast to stop the polio virus. Soon, he discovered why he was successful. “Another paper at the time showed that polio virus translation is quite different from cellular protein synthesis,” says Das. The polio virus, he learned, shuts off its host cell’s protein synthesis but uses a different mechanism, called internal initiation of translation, to make its own proteins. The small RNA from yeast in his experiment was, he realised, somehow preventing the polio virus from using this mechanism.
Some years later, in 1998, Das joined IISc’s Department of Microbiology and Cell Biology. When he started his own lab, Das zeroed in on another virus, the one that causes hepatitis C, which he felt might be vulnerable to a similar approach. Since then, Das and collaborators have developed antivirals that attack various stages of the hepatitis C virus (HCV) lifecycle, along with mechanisms for their targeted delivery to the liver. Much of this work has been done as part of a multidisciplinary Centre of Excellence for Hepatitis C Virus Research, a collaborative effort involving nine labs across India, that he heads.
Das and collaborators have developed antivirals that attack various stages of the hepatitis C virus lifecycle, along with mechanisms for their targeted delivery to the liver
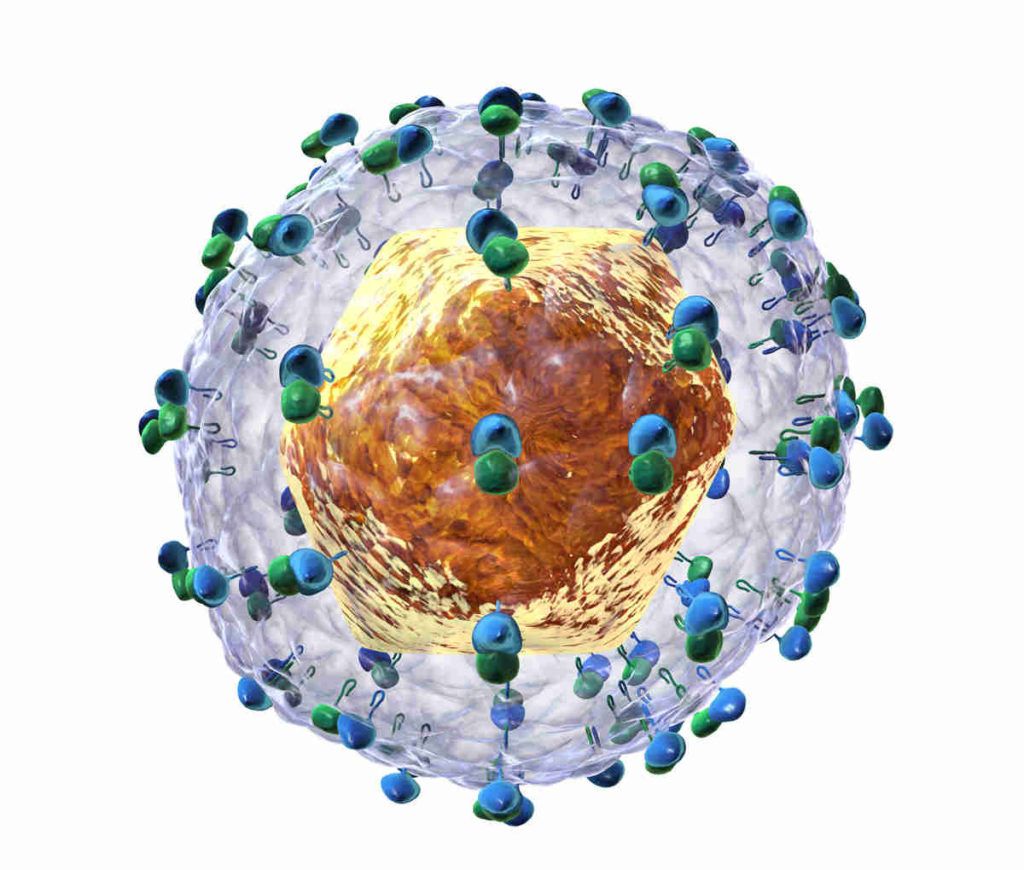
Hepatitis, a disease of the liver, can be due to many causes, including viruses. There are five hepatitis-causing viruses. One of these, the hepatitis C, discovered in 1989, infects an estimated 175 million people worldwide, with 3-4 million persons infected each year through contact with infected blood, via infected needles or blood transfusions. The immune response to HCV infection results in inflammation of liver tissue, which could lead to liver cell damage and hepatocellular carcinoma (liver cancer). In 80% of the patients, HCV causes chronic infection. In India, approximately 15-20% of cases of chronic liver diseases are caused by HCV infection.
There is no vaccine available yet for HCV. Developing antiviral therapies, meanwhile, is a challenge because the RNA virus mutates quickly, leading to variants that may be immune to the antiviral.
An important attribute of an antiviral agent is that it must be specific – it should inhibit only the virus and should leave the host cells unaffected so as to avoid side effects. Das found that for HCV, a key to developing an antiviral agent lies in the way the virus translates its genetic information to make proteins. HCV, like other viruses, hijacks the host cell’s machinery for this. In any cell, the ribosome is the organelle that translates genetic information to synthesise proteins. Once HCV has entered a cell, it tries to use the ribosome to synthesise its proteins. However, as with the polio virus, the way HCV translates its RNA is different from how its host cell does it – the ribosome binds not to the end of the viral RNA, but to an internal sequence. The virus has evolved in such a way that it has less competition with cellular RNA to bind to the ribosome. “It’s as if somebody is entering through the back gate,” says Das. Antiviral agents inhibiting this translation – specifically, the ribosome- HCV RNA interaction – offered a way of stopping the viral RNA replication. What’s more, this mechanism is common to all strains of HCV.
The way HCV translates its RNA is different from how its host cell does it – the ribosome binds not to the end of the viral RNA, but to an internal sequence

Over the years, Das has successfully demonstrated various approaches to inhibiting translation of HCV. One method is to use a small RNA derived from the HCV RNA. “If we use this small RNA as a competitor, it will sequester the host factor and the virus cannot exploit that for protein synthesis,” says Das. With collaborators, he also found success with another approach, one that used one of the virus’ own proteins against it. The virus, once it enters the cell, cannot keep making proteins lest it be caught by the host’s immune system. “They [the virus] have to have a regulation. After they have a little bit of synthesis, they try to stop,” says Das. “One of the viral proteins does this job. It’s like a feedback mechanism.” This particular protein of the virus was identified, a peptide was derived from the RNA-binding region of this protein, and this was found to prevent the viral RNA translation. This is because the peptide locks the binding site on the ribosome, preventing the viral RNA from binding. This interaction was studied by N Srinivasan (Molecular Biophysics Unit) using computational modelling of the structures involved.
HCV infects only humans and chimpanzees; it does not infect mice, meaning that studying HCV infection in animals to understand the host-virus interactions and to develop drugs has been difficult. Now, however, there are “chimeric” mice available which carry a transplanted human liver (their own liver being compromised), which can be infected to study HCV and to test treatments. In such chimeric mice, the above treatments have proved effective.
“So on one hand, even if the virus has entered the cell, you try to prevent their protein synthesis. The other strategy is to block their entry into the cell, using monoclonal antibodies,” says Das. Of the people infected with HCV, about 15% clear the infection on their own. Das’ colleague in the Department, MS Shaila (now retired) and Anjali Karande (Department of Biochemistry), isolated the antibodies that such people develop and made similar antibodies in the lab, called monoclonal antibodies. This was found to be effective in preventing HCV from entering the cells in the first place.
Das has also been working with collaborators in other departments at IISc to develop methods that can deliver these drugs selectively to the liver, to minimise the potential side effects from toxicity. The idea is to use a carrier that can bind to the drug molecules, carry it to the liver, and bind to the appropriate receptor on the target cells. The carrier is sensitive to the pH (a measure of acidicity) of the environment it finds itself in, and can be designed such that the drug molecule that it captured at a certain pH gets released at a different pH. One approach for liver-targeted drug delivery, which N Jayaraman (Department of Organic Chemistry) and Ajay Sood (Department of Physics) have developed, is to use dendrimers. (Dendrimers are polymers that have a branching, tree-like structure.) They have used a class of dendrimers known as PETIM dendrimers, which is able to form a complex with drug molecules via electrostatic interactions. The dendrimers were functionalized to specifically target liver cells.
This has also been studied using simulations by Prabal Maiti (Department of Physics). Another approach is to carry the drug molecule in a “cage” made of polyelectrolyte nanocapsules, which Ashok Raichur (Department of Materials Engineering) is working on.
All this work was on antivirals that can either stop HCV from entering cells or stop its translation if it has already entered. But Das and collaborators have also worked on stopping the virus in the next step in its lifecycle inside a cell – its replication. Following a government initiative to search through herbal compounds for potential treatments for various diseases, Das’ lab began screening natural products for their antiviral action against HCV. They found that the peel of pomegranate (Punica granatum), a fruit long known for its health benefits, especially for the liver, had compounds known as the ellagitannins, which inhibit virus replication. Existing antiviral treatments, even if they treat the hepatitis, can leave behind a damaged liver. “Even after six months of treatment, and even if the patient has completely cleared the virus and recovered, they may still have liver damage which can lead to hepatocellular carcinoma,” says Das. The compound identified in pomegranate, however, was found to protect and heal the liver. Das’ lab had earlier identified a similar compound in extracts from the root and leaf of Phyllanthus amarus, a type of amla (gooseberry), which was also found to inhibit HCV viral enzymes and replication. “This bhui amla is traditional folk medicine that people have used for jaundice and hepatitis,” he says.
They found that the peel of pomegranate (Punica granatum), a fruit long known for its health benefits, especially for the liver, had compounds known as the ellagitannins, which inhibit virus replication

“Anything of this sort has to go through clinical trials,” says Das, “to ensure there are no long-term side effects.” After the paper on the HCV-fighting abilities of pomegranate was published, Das received phone calls from people with hepatitis asking if they should drink pomegranate juice, and how much. “I told them this is an observation. All I can say is that drinking one glass of pomegranate juice is not going to hurt you. But don’t drink ten glasses a day,” he says. “Also have consultations with your clinician, who will know what else you are taking.”
This roadmap – finding a unique mechanism in the virus translation which can be targeted – could be used for developing treatments for other virus infections too. “Research on translation to translational research – that is my research theme,” says Das. “All this was possible because of the untiring efforts and the wonderful teamwork of my students and postdocs over the years, and also at other labs in the HCV-Centre of Excellence,” he adds. “We started with protein synthesis, which is called translation, and ended up with translational research.” He is now looking to team up with partners in industry to further this research and commercialise it.